Itching and Atopy Relief using Azulene Derivatives and High-Content Ceramide Skin Barrier Nano-Liposome Structures by Dry Skin
Keywords:
Clinical study, Itching relief, Guaiazulene, Ceramide, CosmeticsAbstract
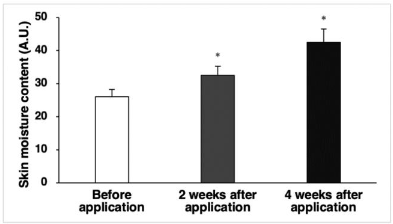
Atopic dermatitis (AD), a chronic inflammatory skin disease, has increased in industrialized nations, impacting approximately 15 to 20% of children and 1 to 3% of adults worldwide. Previous studies suggest that the impairment of the skin barrier and immunological dysfunction can be considered as the two main factors involved in AD. In this study, a balm formulation containing 0.02% guaiazulene, an azulene derivative, and 1.0% high-content ceramide skin barrier nano-liposome structure was developed to effectively treat AD by normalizing these main factors, and clinical trials were conducted with 23 subjects. Compared to before using the product, (1) the skin moisture content in the forearm area increased significantly (p<0.05) by 25.20% and 63.61% after 2 weeks and 4 weeks of use of the product, respectively, (2) the amount of skin moisture loss in the forearm area decreased significantly (p<0.05) by 12.04% and 17.79% after using the product for 2 weeks and 4 weeks, respectively, (3) the skin texture (roughness) of the forearm area significantly decreased (p<0.05) by 3.00% and 4.18% after 2 weeks and 4 weeks of use, respectively, (4) the degree of itchiness due to dry skin significantly decreased (p<0.05) by 47.91% after 2 weeks of use and 69.71% after 4 weeks of use, and the degree of sleep disturbance due to itching caused by dry skin significantly decreased (p<0.05) by 46.22% after using the product for 2 weeks and by 74.07% after using the product for 4 weeks. This study confirmed the outstanding clinical effects of the formulation containing guaiazulene and ceramide on itching relief caused by dry skin.
Downloads
References
Brüssow, H., Turning the inside out: the microbiology of atopic dermatitis. Environmental microbiology, 18 (2016), 2089-2102.
Chang, A. L. S., Chen, S. C., Osterberg, L., Brandt, S., von Grote, E. C., and Meckfessel, M. H., A daily skincare regimen with a unique ceramide and filaggrin formulation rapidly improves chronic xerosis, pruritus, and quality of life in older adults. Geriatric nursing, 39 (2018), 24-28.
EL-YAHYAOUI, A., & OMARY, F. (2022). A Like ELGAMAL Cryptosystem But Resistant To Post-Quantum Attacks. International Journal of Communication Networks and Information Security (IJCNIS), 14(1). https://doi.org/10.17762/ijcnis.v14i1.5180
Silverberg, N. B. and Silverberg, J. I., Inside out or outside in: does atopic dermatitis disrupt barrier function or does disruption of barrier function trigger atopic dermatitis. Cutis, 96 (2015), 359-361.
Avena-Woods, C., Overview of atopic dermatitis. The American journal of managed care, 23 (2017), S115-S123.
AvenaGuttman-Yassky, E., Waldman, A., Ahluwalia, J., Ong, P. Y., and Eichenfield, L. F., Atopic dermatitis: pathogenesis. Seminars in cutaneous medicine and surgery, 36 (2017), 100-103.
Patrick, G. J., Archer, N. K., and Miller, L. S., Which way do we go? Complex interactions in atopic dermatitis pathogenesis. Journal of investigative dermatology, 141 (2021), 274-284.
Park, S. I., Lee, K. W., Park, S., and Shin, M. S., In vitro biological activities of azulene, guaiazulene, and sodium guaiazulene sulfonate and its application to formulations through PEG-PCL micelles. Review of international geographical education online, 11 (2021), 2084-2091.
Phutim-Mangkhalthon, A., Teerakapong, A., Tippayawat, P., Morales, N. P., Morkmued, S., Puasiri, S., ... and Damrongrungruang, T., Anti-inflammatory effect of photodynamic therapy using guaiazulene and red lasers on peripheral blood mononuclear cells. Photodiagnosis and photodynamic therapy, 31 (2020), 101747.
Gunes, T., Akin, M. A., Sarici, D., Hallac, K., Kurtoglu, S., and Hashimoto, T., Guaiazulene: a new treatment option for recalcitrant diaper dermatitis in NICU patients. The journal of maternal-fetal & neonatal medicine, 26 (2013), 197-200.
Wertz, P. W. and Downing, D. T., Ceramides of pig epidermis: structure determination. Journal of lipid research, 24 (1983), 759-765.
Choi, M. J. and Maibach, H. I., Role of ceramides in barrier function of healthy and diseased skin. American journal of clinical dermatology, 6 (2005), 215-223.
Gomez-Munoz, A., Presa, N., Gomez-Larrauri, A., Rivera, I. G., Trueba, M., and Ordonez, M., Control of inflammatory responses by ceramide, sphingosine 1-phosphate and ceramide 1-phosphate. Progress in lipid research, 61 (2016), 51-62.
Fischer, C. L., Drake, D. R., Dawson, D. V., Blanchette, D. R., Brogden, K. A., and Wertz, P. W., Antibacterial activity of sphingoid bases and fatty acids against Gram-positive and Gram-negative bacteria. Antimicrobial agents and chemotherapy, 56 (2012), 1157-1161.
André Sanches Fonseca Sobrinho. (2020). An Embedded Systems Remote Course. Journal of Online Engineering Education, 11(2), 01–07. Retrieved from http://onlineengineeringeducation.com/index.php/joee/article/view/39
Cho, S. H., Strickland, I., Tomkinson, A., Fehringer, A. P., Gelfand, E. W., and Leung, D. Y., Preferential binding of Staphylococcus aureus to skin sites of Th2-mediated inflammation in a murine model. Journal of investigative dermatology, 116 (2001), 658-663.
Ye, L., Wang, Z., Li, Z., Lv, C., and Man, M. Q., Validation of GPS kin Barrier® for assessing epidermal permeability barrier function and stratum corneum hydration in humans. Skin research and technology, 25 (2019), 25-29.
Roskos, K. V. and Guy, R. H., Assessment of skin barrier function using transepidermal water loss: effect of age. Pharmaceutical research, 6 (1989), 949-953.
Misra, M. and Ananthapadmanabhan, K. P., Changes in epidermis and transepidermal water loss. Journal of the society of cosmetic chemists, 48 (1997), 219-234.
Krishna, P. R. ., and P. . Rajarajeswari. “EapGAFS: Microarray Dataset for Ensemble Classification for Diseases Prediction”. International Journal on Recent and Innovation Trends in Computing and Communication, vol. 10, no. 8, Aug. 2022, pp. 01-15, doi:10.17762/ijritcc.v10i8.5664.
Barel, A. O. and Clarys, P., Study of the stratum corneum barrier function by transepidermal water loss measurements: comparison between two commercial instruments: evaporimeter® and Tewameter®. Skin pharmacology and physiology, 8 (1995), 186-195.
Chawla, A. (2022). Phishing website analysis and detection using Machine Learning. International Journal of Intelligent Systems and Applications in Engineering, 10(1), 10–16. https://doi.org/10.18201/ijisae.2022.262
Tagami, H., Location‐related differences in structure and function of the stratum corneum with special emphasis on those of the facial skin. International journal of cosmetic science, 30 (2008), 413-434.
Chamlin, S. L., Mattson, C. L., Frieden, I. J., Williams, M. L., Mancini, A. J., Cella, D., and Chren, M. M., The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Archives of pediatrics & adolescent medicine, 159 (2005), 745-750.
Iwabuchi, K., Nakayama, H., Oizumi, A., Suga, Y., Ogawa, H., and Takamori, K., Role of ceramide from glycosphingolipids and its metabolites in immunological and inflammatory responses in humans. Mediators of inflammation, 2015 (2015).
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
All papers should be submitted electronically. All submitted manuscripts must be original work that is not under submission at another journal or under consideration for publication in another form, such as a monograph or chapter of a book. Authors of submitted papers are obligated not to submit their paper for publication elsewhere until an editorial decision is rendered on their submission. Further, authors of accepted papers are prohibited from publishing the results in other publications that appear before the paper is published in the Journal unless they receive approval for doing so from the Editor-In-Chief.
IJISAE open access articles are licensed under a Creative Commons Attribution-ShareAlike 4.0 International License. This license lets the audience to give appropriate credit, provide a link to the license, and indicate if changes were made and if they remix, transform, or build upon the material, they must distribute contributions under the same license as the original.





